Jump to navigation
Jump to search
NCBI: 03-AUG-2016
⊟Summary[edit | edit source]
- organism: Staphylococcus aureus NCTC8325
- locus tag: SAOUHSC_01199
- pan locus tag?: SAUPAN003515000
- symbol: SAOUHSC_01199
- pan gene symbol?: fabG
- synonym:
- product: 3-oxoacyl-(acyl-carrier-protein) reductase
⊟Genome View[edit | edit source]
⊟Gene[edit | edit source]
⊟General[edit | edit source]
⊟Accession numbers[edit | edit source]
- Gene ID: 3919331 NCBI
- RefSeq: YP_499737 NCBI
- BioCyc: G1I0R-1124 BioCyc
- MicrobesOnline: 1289651 MicrobesOnline
⊟Phenotype[edit | edit source]
Share your knowledge and add information here. [edit]
⊟DNA sequence[edit | edit source]
- 1
61
121
181
241
301
361
421
481
541
601
661
721ATGACTAAGAGTGCTTTAGTAACAGGTGCATCAAGAGGAATTGGACGTAGTATTGCGTTA
CAATTAGCAGAAGAAGGATATAATGTAGCAGTAAACTATGCAGGCAGCAAAGAGAAAGCT
GAAGCAGTAGTCGAAGAAATCAAAGCTAAAGGTGTTGACAGTTTTGCGATTCAAGCAAAT
GTTGCCGATGCTGATGAAGTTAAAGCAATGATTAAAGAAGTAGTTAGCCAATTTGGTTCT
TTAGATGTTTTAGTAAATAATGCAGGTATTACTCGCGATAATTTATTAATGCGTATGAAA
GAACAAGAGTGGGATGATGTTATTGACACAAACTTAAAAGGTGTATTTAACTGTATCCAA
AAAGCAACACCACAAATGTTAAGACAACGTAGTGGTGCTATCATCAATTTATCAAGTGTT
GTTGGAGCAGTAGGTAATCCGGGACAAGCAAACTATGTTGCAACAAAAGCAGGTGTTATT
GGTTTAACTAAATCTGCGGCGCGTGAATTAGCATCTCGTGGTATCACTGTAAATGCAGTT
GCACCTGGTTTTATTGTTTCTGATATGACAGATGCTTTAAGTGATGAGCTTAAAGAACAA
ATGTTGACTCAAATTCCGTTAGCACGTTTTGGTCAAGACACAGATATTGCTAATACAGTA
GCGTTCTTAGCATCAGACAAAGCAAAATATATTACAGGTCAAACAATCCATGTAAATGGT
GGAATGTACATGTAA60
120
180
240
300
360
420
480
540
600
660
720
735
⊟Protein[edit | edit source]
⊟General[edit | edit source]
- locus tag: SAOUHSC_01199
- symbol: SAOUHSC_01199
- description: 3-oxoacyl-(acyl-carrier-protein) reductase
- length: 244
- theoretical pI: 5.10093
- theoretical MW: 25886.4
- GRAVY: 0.0172131
⊟Function[edit | edit source]
- reaction: EC 1.1.1.100? ExPASy3-oxoacyl-[acyl-carrier-protein] reductase (3R)-3-hydroxyacyl-[acyl-carrier-protein] + NADP+ = 3-oxoacyl-[acyl-carrier-protein] + NADPH
- TIGRFAM: Fatty acid and phospholipid metabolism Biosynthesis 3-oxoacyl-[acyl-carrier-protein] reductase (TIGR01830; EC 1.1.1.100; HMM-score: 362.9)and 19 moreacetoacetyl-CoA reductase (TIGR01829; EC 1.1.1.36; HMM-score: 284.5)Fatty acid and phospholipid metabolism Biosynthesis putative 3-oxoacyl-(acyl-carrier-protein) reductase (TIGR01831; HMM-score: 242.1)3-hydroxybutyrate dehydrogenase (TIGR01963; HMM-score: 207)Energy metabolism Fermentation acetoin reductases (TIGR02415; EC 1.1.1.-; HMM-score: 206.7)Unknown function Enzymes of unknown specificity SDR family mycofactocin-dependent oxidoreductase (TIGR03971; EC 1.1.99.-; HMM-score: 204.9)Energy metabolism Biosynthesis and degradation of polysaccharides 2-deoxy-D-gluconate 3-dehydrogenase (TIGR01832; EC 1.1.1.125; HMM-score: 197.2)2-hydroxycyclohexanecarboxyl-CoA dehydrogenase (TIGR03206; EC 1.1.1.-; HMM-score: 167.2)2,3-dihydro-2,3-dihydroxybenzoate dehydrogenase (TIGR04316; EC 1.3.1.28; HMM-score: 162.7)Unknown function Enzymes of unknown specificity SDR family mycofactocin-dependent oxidoreductase (TIGR04504; EC 1.1.99.-; HMM-score: 140.8)pteridine reductase (TIGR02685; EC 1.5.1.33; HMM-score: 131.8)rhamnulose-1-phosphate aldolase/alcohol dehydrogenase (TIGR02632; EC 1.1.1.1,4.1.2.19; HMM-score: 117.4)cis-2,3-dihydrobiphenyl-2,3-diol dehydrogenase (TIGR03325; EC 1.3.1.56; HMM-score: 90.5)sepiapterin reductase (TIGR01500; EC 1.1.1.153; HMM-score: 35.7)Biosynthesis of cofactors, prosthetic groups, and carriers Chlorophyll and bacteriochlorphyll light-dependent protochlorophyllide reductase (TIGR01289; EC 1.3.1.33; HMM-score: 23.1)methylmalonyl-CoA mutase C-terminal domain (TIGR00640; HMM-score: 17.8)Energy metabolism Sugars UDP-glucose 4-epimerase GalE (TIGR01179; EC 5.1.3.2; HMM-score: 15)NAD dependent epimerase/dehydratase, LLPSF_EDH_00030 family (TIGR04180; HMM-score: 13.3)Transport and binding proteins Carbohydrates, organic alcohols, and acids tricarboxylate carrier (TIGR00798; HMM-score: 11.8)Biosynthesis of cofactors, prosthetic groups, and carriers Thiamine glycine oxidase ThiO (TIGR02352; EC 1.4.3.19; HMM-score: 11.4)
- TheSEED :
- 3-oxoacyl-[acyl-carrier protein] reductase (EC 1.1.1.100)
- PFAM: NADP_Rossmann (CL0063) adh_short_C2; Enoyl-(Acyl carrier protein) reductase (PF13561; HMM-score: 239.5)adh_short; short chain dehydrogenase (PF00106; HMM-score: 222.8)and 12 moreKR; KR domain (PF08659; HMM-score: 84.4)Epimerase; NAD dependent epimerase/dehydratase family (PF01370; HMM-score: 23.4)SDR; SDR-like rossmann domain (PF23441; HMM-score: 22.5)PDDEXK (CL0236) HFX_2341_N; HFX_2341-like, N-terminal (PF19810; HMM-score: 15.2)HTH (CL0123) MCM_WH_arc; Archaeal MCM, winged-helix (PF21120; HMM-score: 14)NADP_Rossmann (CL0063) NmrA; NmrA-like family (PF05368; HMM-score: 13.7)no clan defined B12-binding; B12 binding domain (PF02310; HMM-score: 13.3)SPOR; SPOR domain (PF05036; HMM-score: 13)DUF7336; Domain of unknown function (DUF7336) (PF24024; HMM-score: 12.9)NADP_Rossmann (CL0063) GDP_Man_Dehyd; GDP-mannose 4,6 dehydratase (PF16363; HMM-score: 12.8)NAD_binding_10; NAD(P)H-binding (PF13460; HMM-score: 12.3)CoA_binding_2; CoA binding domain (PF13380; HMM-score: 9.4)
⊟Structure, modifications & cofactors[edit | edit source]
- domains:
- modifications:
- cofactors:
- effectors:
⊟Localization[edit | edit source]
- PSORTb: Cytoplasmic
- Cytoplasmic Score: 9.97
- Cytoplasmic Membrane Score: 0
- Cellwall Score: 0.01
- Extracellular Score: 0.02
- Internal Helices: 0
- DeepLocPro: Cytoplasmic
- Cytoplasmic Score: 0.9008
- Cytoplasmic Membrane Score: 0.06
- Cell wall & surface Score: 0.0114
- Extracellular Score: 0.0278
- LocateP: Intracellular
- Prediction by SwissProt Classification: Cytoplasmic
- Pathway Prediction: No pathway
- Intracellular possibility: 1
- Signal peptide possibility: -1
- N-terminally Anchored Score: 1
- Predicted Cleavage Site: No CleavageSite
- SignalP: no predicted signal peptide
- SP(Sec/SPI): 0.019676
- TAT(Tat/SPI): 0.002441
- LIPO(Sec/SPII): 0.003352
- predicted transmembrane helices (TMHMM): 0
⊟Accession numbers[edit | edit source]
⊟Protein sequence[edit | edit source]
- MTKSALVTGASRGIGRSIALQLAEEGYNVAVNYAGSKEKAEAVVEEIKAKGVDSFAIQANVADADEVKAMIKEVVSQFGSLDVLVNNAGITRDNLLMRMKEQEWDDVIDTNLKGVFNCIQKATPQMLRQRSGAIINLSSVVGAVGNPGQANYVATKAGVIGLTKSAARELASRGITVNAVAPGFIVSDMTDALSDELKEQMLTQIPLARFGQDTDIANTVAFLASDKAKYITGQTIHVNGGMYM
⊟Experimental data[edit | edit source]
- experimentally validated: PeptideAtlas [2] [3]
- protein localization: data available for COL
- quantitative data / protein copy number per cell: data available for COL
- interaction partners:
SAOUHSC_02512 (rplC) 50S ribosomal protein L3 [4] (data from MRSA252) SAOUHSC_02494 (rpsE) 30S ribosomal protein S5 [4] (data from MRSA252) SAOUHSC_00529 elongation factor G [4] (data from MRSA252) SAOUHSC_00530 elongation factor Tu [4] (data from MRSA252) SAOUHSC_01040 pyruvate dehydrogenase complex, E1 component subunit alpha [4] (data from MRSA252) SAOUHSC_01490 DNA-binding protein HU [4] (data from MRSA252) SAOUHSC_02486 30S ribosomal protein S11 [4] (data from MRSA252)
⊟Expression & Regulation[edit | edit source]
⊟Operon[edit | edit source]
- MicrobesOnline: SAOUHSC_01196 > SAOUHSC_01197 > SAOUHSC_01198 > SAOUHSC_01199predicted SigA promoter [5] : SAOUHSC_01194 > S493 > SAOUHSC_01196 > SAOUHSC_01197 > SAOUHSC_01198 > SAOUHSC_01199 > S494 > S495 > acpPpredicted SigA promoter [5] : SAOUHSC_01196 > SAOUHSC_01197 > SAOUHSC_01198 > SAOUHSC_01199 > S494 > S495 > acpP > S496 > rnc > S497 > SAOUHSC_01204 > SAOUHSC_01205 > SAOUHSC_01206 > SAOUHSC_01207 > S498
⊟Regulation[edit | edit source]
- regulator: FapR* (repression) regulon
FapR* (TF) important in Fatty acid biosynthesis; RegPrecise transcription unit transferred from N315 data RegPrecise
⊟Transcription pattern[edit | edit source]
- S.aureus Expression Data Browser: [5]
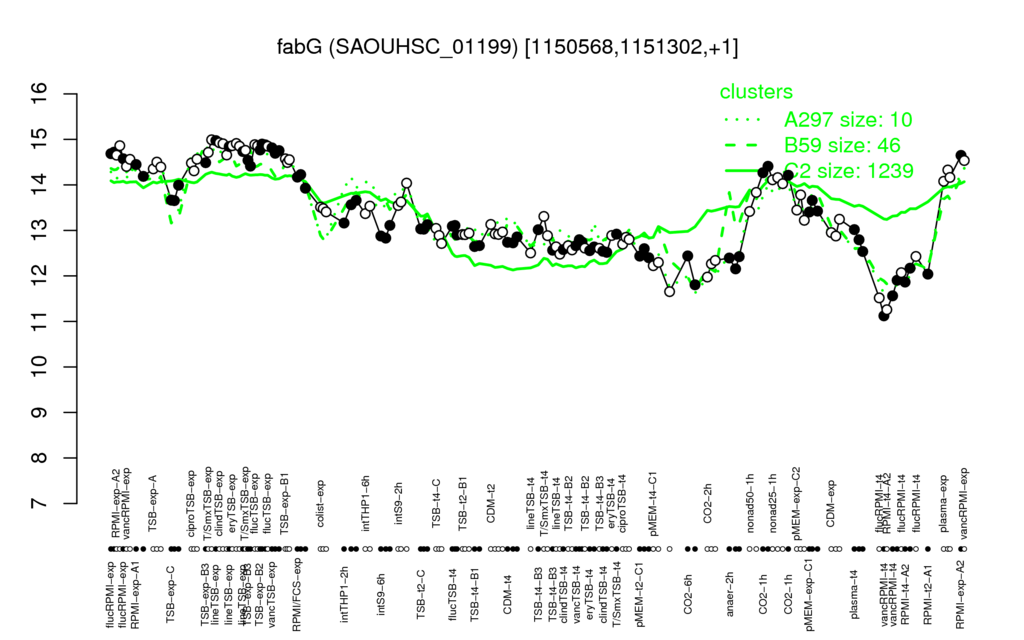 Multi-gene expression profiles
Multi-gene expression profiles
⊟Protein synthesis (provided by Aureolib)[edit | edit source]
⊟Protein stability[edit | edit source]
- half-life: no data available
⊟Biological Material[edit | edit source]
⊟Mutants[edit | edit source]
⊟Expression vector[edit | edit source]
⊟lacZ fusion[edit | edit source]
⊟GFP fusion[edit | edit source]
⊟two-hybrid system[edit | edit source]
⊟FLAG-tag construct[edit | edit source]
⊟Antibody[edit | edit source]
⊟Other Information[edit | edit source]
You can add further information about the gene and protein here. [edit]
⊟Literature[edit | edit source]
⊟References[edit | edit source]
- ↑ Roy R Chaudhuri, Andrew G Allen, Paul J Owen, Gil Shalom, Karl Stone, Marcus Harrison, Timothy A Burgis, Michael Lockyer, Jorge Garcia-Lara, Simon J Foster, Stephen J Pleasance, Sarah E Peters, Duncan J Maskell, Ian G Charles
Comprehensive identification of essential Staphylococcus aureus genes using Transposon-Mediated Differential Hybridisation (TMDH).
BMC Genomics: 2009, 10;291
[PubMed:19570206] [WorldCat.org] [DOI] (I e) - ↑ Maren Depke, Stephan Michalik, Alexander Rabe, Kristin Surmann, Lars Brinkmann, Nico Jehmlich, Jörg Bernhardt, Michael Hecker, Bernd Wollscheid, Zhi Sun, Robert L Moritz, Uwe Völker, Frank Schmidt
A peptide resource for the analysis of Staphylococcus aureus in host-pathogen interaction studies.
Proteomics: 2015, 15(21);3648-61
[PubMed:26224020] [WorldCat.org] [DOI] (I p) - ↑ Stephan Michalik, Maren Depke, Annette Murr, Manuela Gesell Salazar, Ulrike Kusebauch, Zhi Sun, Tanja C Meyer, Kristin Surmann, Henrike Pförtner, Petra Hildebrandt, Stefan Weiss, Laura Marcela Palma Medina, Melanie Gutjahr, Elke Hammer, Dörte Becher, Thomas Pribyl, Sven Hammerschmidt, Eric W Deutsch, Samuel L Bader, Michael Hecker, Robert L Moritz, Ulrike Mäder, Uwe Völker, Frank Schmidt
A global Staphylococcus aureus proteome resource applied to the in vivo characterization of host-pathogen interactions.
Sci Rep: 2017, 7(1);9718
[PubMed:28887440] [WorldCat.org] [DOI] (I e) - ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 Artem Cherkasov, Michael Hsing, Roya Zoraghi, Leonard J Foster, Raymond H See, Nikolay Stoynov, Jihong Jiang, Sukhbir Kaur, Tian Lian, Linda Jackson, Huansheng Gong, Rick Swayze, Emily Amandoron, Farhad Hormozdiari, Phuong Dao, Cenk Sahinalp, Osvaldo Santos-Filho, Peter Axerio-Cilies, Kendall Byler, William R McMaster, Robert C Brunham, B Brett Finlay, Neil E Reiner
Mapping the protein interaction network in methicillin-resistant Staphylococcus aureus.
J Proteome Res: 2011, 10(3);1139-50
[PubMed:21166474] [WorldCat.org] [DOI] (I p) - ↑ 5.0 5.1 5.2 Ulrike Mäder, Pierre Nicolas, Maren Depke, Jan Pané-Farré, Michel Debarbouille, Magdalena M van der Kooi-Pol, Cyprien Guérin, Sandra Dérozier, Aurelia Hiron, Hanne Jarmer, Aurélie Leduc, Stephan Michalik, Ewoud Reilman, Marc Schaffer, Frank Schmidt, Philippe Bessières, Philippe Noirot, Michael Hecker, Tarek Msadek, Uwe Völker, Jan Maarten van Dijl
Staphylococcus aureus Transcriptome Architecture: From Laboratory to Infection-Mimicking Conditions.
PLoS Genet: 2016, 12(4);e1005962
[PubMed:27035918] [WorldCat.org] [DOI] (I e)
⊟Relevant publications[edit | edit source]
Debajyoti Dutta, Sudipta Bhattacharyya, Amit Kumar Das
Crystal structure and fluorescence studies reveal the role of helical dimeric interface of staphylococcal FabG1 in positive cooperativity for NADPH.
Proteins: 2012, 80(4);1250-7
[PubMed:22275129] [WorldCat.org] [DOI] (I p)
