⊟Summary[edit | edit source]
- organism: Staphylococcus aureus NCTC8325
- locus tag: SAOUHSC_00790
- pan locus tag?: SAUPAN002695000
- symbol: clpP
- pan gene symbol?: clpP
- synonym:
- product: ATP-dependent Clp protease proteolytic subunit
⊟Genome View[edit | edit source]
⊟Gene[edit | edit source]
⊟General[edit | edit source]
⊟Accession numbers[edit | edit source]
- Gene ID: 3919354 NCBI
- RefSeq: YP_499347 NCBI
- BioCyc: G1I0R-740 BioCyc
- MicrobesOnline: 1289258 MicrobesOnline
⊟Phenotype[edit | edit source]
Share your knowledge and add information here. [edit]
⊟DNA sequence[edit | edit source]
- 1
61
121
181
241
301
361
421
481
541ATGAATTTAATTCCTACAGTTATTGAAACAACAAACCGCGGTGAACGTGCATATGATATA
TACTCACGTTTATTAAAAGACCGTATTATTATGTTAGGTTCACAAATTGATGACAACGTA
GCAAATTCAATCGTATCACAGTTATTATTCTTACAAGCGCAAGACTCAGAGAAAGATATT
TATTTATACATTAATTCACCAGGTGGAAGTGTAACAGCTGGTTTTGCGATTTATGATACA
ATTCAACACATTAAACCTGATGTTCAAACAATTTGTATCGGTATGGCTGCATCAATGGGA
TCATTCTTATTAGCAGCTGGTGCAAAAGGTAAACGTTTCGCGTTACCAAATGCAGAAGTA
ATGATTCACCAACCATTAGGTGGTGCTCAAGGACAAGCAACTGAAATCGAAATTGCTGCA
AATCACATTTTAAAAACACGTGAAAAATTAAACCGCATTTTATCAGAGCGTACTGGTCAA
AGTATTGAAAAAATACAAAAAGACACAGATCGTGATAACTTCTTAACTGCAGAAGAAGCT
AAAGAATATGGCTTAATTGATGAAGTGATGGTACCTGAAACAAAATAA60
120
180
240
300
360
420
480
540
588
⊟Protein[edit | edit source]
⊟General[edit | edit source]
- locus tag: SAOUHSC_00790
- symbol: ClpP
- description: ATP-dependent Clp protease proteolytic subunit
- length: 195
- theoretical pI: 4.89682
- theoretical MW: 21513.4
- GRAVY: -0.188205
⊟Function[edit | edit source]
- reaction: EC 3.4.21.92? ExPASyEndopeptidase Clp Hydrolysis of proteins to small peptides in the presence of ATP and magnesium. Alpha-casein is the usual test substrate. In the absence of ATP, only oligopeptides shorter than five residues are hydrolyzed (such as succinyl-Leu-Tyr-|-NHMec; and Leu-Tyr-Leu-|-Tyr-Trp, in which cleavage of the -Tyr-|-Leu- and -Tyr-|-Trp bonds also occurs)
- TIGRFAM: Protein fate Degradation of proteins, peptides, and glycopeptides ATP-dependent Clp endopeptidase, proteolytic subunit ClpP (TIGR00493; EC 3.4.21.92; HMM-score: 346.7)and 3 moreProtein fate Degradation of proteins, peptides, and glycopeptides signal peptide peptidase SppA, 36K type (TIGR00706; EC 3.4.-.-; HMM-score: 28.4)Protein fate Degradation of proteins, peptides, and glycopeptides signal peptide peptidase SppA, 67K type (TIGR00705; EC 3.4.-.-; HMM-score: 21.7)Energy metabolism Other 4-hydroxy-2-oxovalerate aldolase (TIGR03217; EC 4.1.3.39; HMM-score: 12.9)
- TheSEED :
- ATP-dependent Clp protease proteolytic subunit ClpP (EC 3.4.21.92)
Protein Metabolism Protein degradation Proteasome bacterial ATP-dependent Clp protease proteolytic subunit (EC 3.4.21.92)and 2 more - PFAM: ClpP_crotonase (CL0127) CLP_protease; Clp protease (PF00574; HMM-score: 301.2)and 2 moreNfeD1b_N; NfeD1b, N-terminal domain (PF25145; HMM-score: 15.5)Peptidase_S49; Peptidase family S49 (PF01343; HMM-score: 12.4)
⊟Structure, modifications & cofactors[edit | edit source]
- domains:
- modifications:
- cofactors:
- effectors:
⊟Localization[edit | edit source]
- PSORTb: Cytoplasmic
- Cytoplasmic Score: 9.67
- Cytoplasmic Membrane Score: 0.01
- Cellwall Score: 0.15
- Extracellular Score: 0.17
- Internal Helices: 0
- DeepLocPro: Cytoplasmic
- Cytoplasmic Score: 0.8898
- Cytoplasmic Membrane Score: 0.0536
- Cell wall & surface Score: 0.002
- Extracellular Score: 0.0545
- LocateP: Intracellular
- Prediction by SwissProt Classification: Cytoplasmic
- Pathway Prediction: No pathway
- Intracellular possibility: 1
- Signal peptide possibility: -1
- N-terminally Anchored Score: -1
- Predicted Cleavage Site: No CleavageSite
- SignalP: no predicted signal peptide
- SP(Sec/SPI): 0.003841
- TAT(Tat/SPI): 0.000397
- LIPO(Sec/SPII): 0.000526
- predicted transmembrane helices (TMHMM): 0
⊟Accession numbers[edit | edit source]
⊟Protein sequence[edit | edit source]
- MNLIPTVIETTNRGERAYDIYSRLLKDRIIMLGSQIDDNVANSIVSQLLFLQAQDSEKDIYLYINSPGGSVTAGFAIYDTIQHIKPDVQTICIGMAASMGSFLLAAGAKGKRFALPNAEVMIHQPLGGAQGQATEIEIAANHILKTREKLNRILSERTGQSIEKIQKDTDRDNFLTAEEAKEYGLIDEVMVPETK
⊟Experimental data[edit | edit source]
- experimentally validated: PeptideAtlas [2] [3]
- protein localization: data available for COL
- quantitative data / protein copy number per cell: data available for COL
- interaction partners:
SAOUHSC_02505 (rplP) 50S ribosomal protein L16 [4] (data from MRSA252) SAOUHSC_01211 (rplS) 50S ribosomal protein L19 [4] (data from MRSA252) SAOUHSC_02965 carbamate kinase [4] (data from MRSA252)
⊟Expression & Regulation[edit | edit source]
⊟Operon[edit | edit source]
⊟Regulation[edit | edit source]
- regulator: CtsR* (repression) regulon
CtsR* (TF) important in Heat shock response; compare RegPrecise for N315 [5]
⊟Transcription pattern[edit | edit source]
- S.aureus Expression Data Browser: [5]
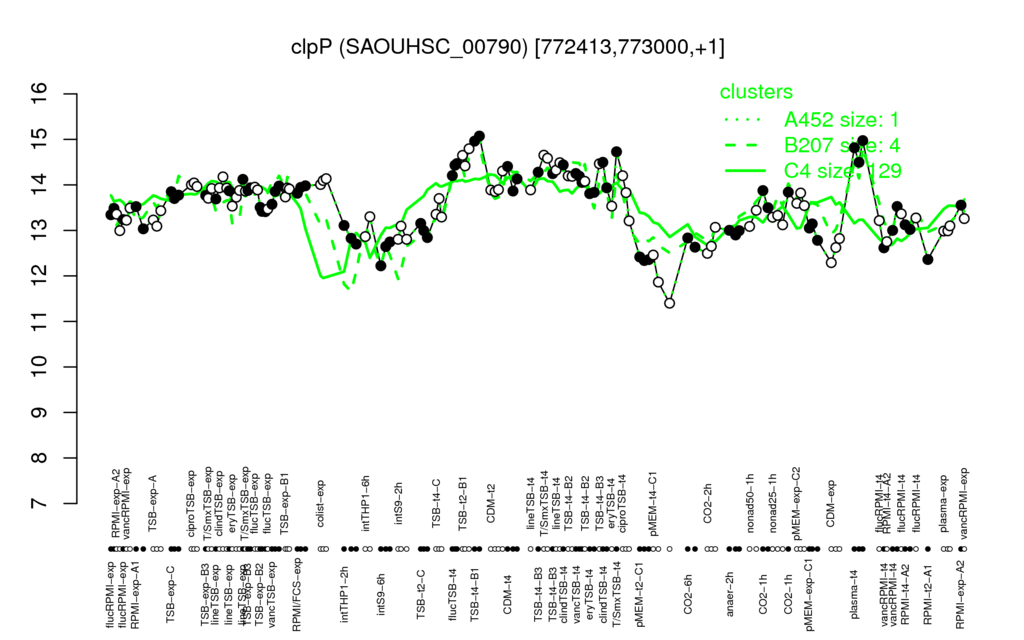 Multi-gene expression profiles
Multi-gene expression profiles
⊟Protein synthesis (provided by Aureolib)[edit | edit source]
⊟Protein stability[edit | edit source]
- half-life: no data available
⊟Biological Material[edit | edit source]
⊟Mutants[edit | edit source]
⊟Expression vector[edit | edit source]
⊟lacZ fusion[edit | edit source]
⊟GFP fusion[edit | edit source]
⊟two-hybrid system[edit | edit source]
⊟FLAG-tag construct[edit | edit source]
⊟Antibody[edit | edit source]
⊟Other Information[edit | edit source]
You can add further information about the gene and protein here. [edit]
⊟Literature[edit | edit source]
⊟References[edit | edit source]
- ↑ Roy R Chaudhuri, Andrew G Allen, Paul J Owen, Gil Shalom, Karl Stone, Marcus Harrison, Timothy A Burgis, Michael Lockyer, Jorge Garcia-Lara, Simon J Foster, Stephen J Pleasance, Sarah E Peters, Duncan J Maskell, Ian G Charles
Comprehensive identification of essential Staphylococcus aureus genes using Transposon-Mediated Differential Hybridisation (TMDH).
BMC Genomics: 2009, 10;291
[PubMed:19570206] [WorldCat.org] [DOI] (I e) - ↑ Maren Depke, Stephan Michalik, Alexander Rabe, Kristin Surmann, Lars Brinkmann, Nico Jehmlich, Jörg Bernhardt, Michael Hecker, Bernd Wollscheid, Zhi Sun, Robert L Moritz, Uwe Völker, Frank Schmidt
A peptide resource for the analysis of Staphylococcus aureus in host-pathogen interaction studies.
Proteomics: 2015, 15(21);3648-61
[PubMed:26224020] [WorldCat.org] [DOI] (I p) - ↑ Stephan Michalik, Maren Depke, Annette Murr, Manuela Gesell Salazar, Ulrike Kusebauch, Zhi Sun, Tanja C Meyer, Kristin Surmann, Henrike Pförtner, Petra Hildebrandt, Stefan Weiss, Laura Marcela Palma Medina, Melanie Gutjahr, Elke Hammer, Dörte Becher, Thomas Pribyl, Sven Hammerschmidt, Eric W Deutsch, Samuel L Bader, Michael Hecker, Robert L Moritz, Ulrike Mäder, Uwe Völker, Frank Schmidt
A global Staphylococcus aureus proteome resource applied to the in vivo characterization of host-pathogen interactions.
Sci Rep: 2017, 7(1);9718
[PubMed:28887440] [WorldCat.org] [DOI] (I e) - ↑ 4.0 4.1 4.2 Artem Cherkasov, Michael Hsing, Roya Zoraghi, Leonard J Foster, Raymond H See, Nikolay Stoynov, Jihong Jiang, Sukhbir Kaur, Tian Lian, Linda Jackson, Huansheng Gong, Rick Swayze, Emily Amandoron, Farhad Hormozdiari, Phuong Dao, Cenk Sahinalp, Osvaldo Santos-Filho, Peter Axerio-Cilies, Kendall Byler, William R McMaster, Robert C Brunham, B Brett Finlay, Neil E Reiner
Mapping the protein interaction network in methicillin-resistant Staphylococcus aureus.
J Proteome Res: 2011, 10(3);1139-50
[PubMed:21166474] [WorldCat.org] [DOI] (I p) - ↑ 5.0 5.1 5.2 5.3 Ulrike Mäder, Pierre Nicolas, Maren Depke, Jan Pané-Farré, Michel Debarbouille, Magdalena M van der Kooi-Pol, Cyprien Guérin, Sandra Dérozier, Aurelia Hiron, Hanne Jarmer, Aurélie Leduc, Stephan Michalik, Ewoud Reilman, Marc Schaffer, Frank Schmidt, Philippe Bessières, Philippe Noirot, Michael Hecker, Tarek Msadek, Uwe Völker, Jan Maarten van Dijl
Staphylococcus aureus Transcriptome Architecture: From Laboratory to Infection-Mimicking Conditions.
PLoS Genet: 2016, 12(4);e1005962
[PubMed:27035918] [WorldCat.org] [DOI] (I e)
⊟Relevant publications[edit | edit source]
Antje Michel, Franziska Agerer, Christof R Hauck, Mathias Herrmann, Joachim Ullrich, Jörg Hacker, Knut Ohlsen
Global regulatory impact of ClpP protease of Staphylococcus aureus on regulons involved in virulence, oxidative stress response, autolysis, and DNA repair.
J Bacteriol: 2006, 188(16);5783-96
[PubMed:16885446] [WorldCat.org] [DOI] (P p)Sebastian R Geiger, Thomas Böttcher, Stephan A Sieber, Patrick Cramer
A conformational switch underlies ClpP protease function.
Angew Chem Int Ed Engl: 2011, 50(25);5749-52
[PubMed:21544912] [WorldCat.org] [DOI] (I p)Malte Gersch, Anja List, Michael Groll, Stephan A Sieber
Insights into structural network responsible for oligomerization and activity of bacterial virulence regulator caseinolytic protease P (ClpP) protein.
J Biol Chem: 2012, 287(12);9484-94
[PubMed:22291011] [WorldCat.org] [DOI] (I p)Jingyuan Feng, Stephan Michalik, Anders N Varming, Julie H Andersen, Dirk Albrecht, Lotte Jelsbak, Stefanie Krieger, Knut Ohlsen, Michael Hecker, Ulf Gerth, Hanne Ingmer, Dorte Frees
Trapping and proteomic identification of cellular substrates of the ClpP protease in Staphylococcus aureus.
J Proteome Res: 2013, 12(2);547-58
[PubMed:23253041] [WorldCat.org] [DOI] (I p)
